You’ve probably noticed how older folks struggle more with infections and take longer to bounce back from sickness. A big part of that boils down to the stem cells in our bone marrow—the ones that crank out all our blood cells—starting to falter over time. Now, researchers from Japan and the U.S. have pinpointed a key mechanism that could explain why these cells lose their mojo as we get older.
Damaged Mitochondria Seem to Drive the Aging of Blood-Making Stem Cells
Hematopoietic stem cells are absolute MVPs in your body. Nestled in the bone marrow, they’re the source of all your major blood cells: red blood cells, white blood cells, and platelets. Plus, they can keep replenishing themselves, so your body always has a fresh supply ready to generate new blood.
When you’re young, this setup runs like clockwork. These stem cells keep your blood supply topped off and strike the right balance across blood cell types. But as the years pile on, things start to go off the rails. The cells lose their stamina, struggle to renew themselves, and skew toward producing just certain blood cell types. The fallout? Your blood and immune systems just don’t perform like they used to.
That’s why a team led by Dr. Masayuki Yamashita at St. Jude Children’s Research Hospital zeroed in on the MLKL protein. For years, it’s been pegged mostly to necroptosis, a form of programmed cell death. But here, its role proved way sneakier. It doesn’t outright kill the cell—instead, it throws a wrench into how the cell operates.
“We observed an unexpected phenomenon in the stem cells of MLKL-deficient mice repeatedly exposed to stress. Age-associated changes were significantly reduced even though we detected no difference in cell death. This led us to ask whether this mechanism might cause functional changes without actually killing the cells,” Dr. Yamashita explains.
This Time, the Cell Death Protein Wasn’t Actually Killing Anything
The researchers tested several groups of mice. Some were regular, others lacked the MLKL protein, and one group was missing the RIPK3 protein. They hit the animals with various stresses designed to mimic real-world aging pressures, like inflammation, replication stress, and cancer-like stress.
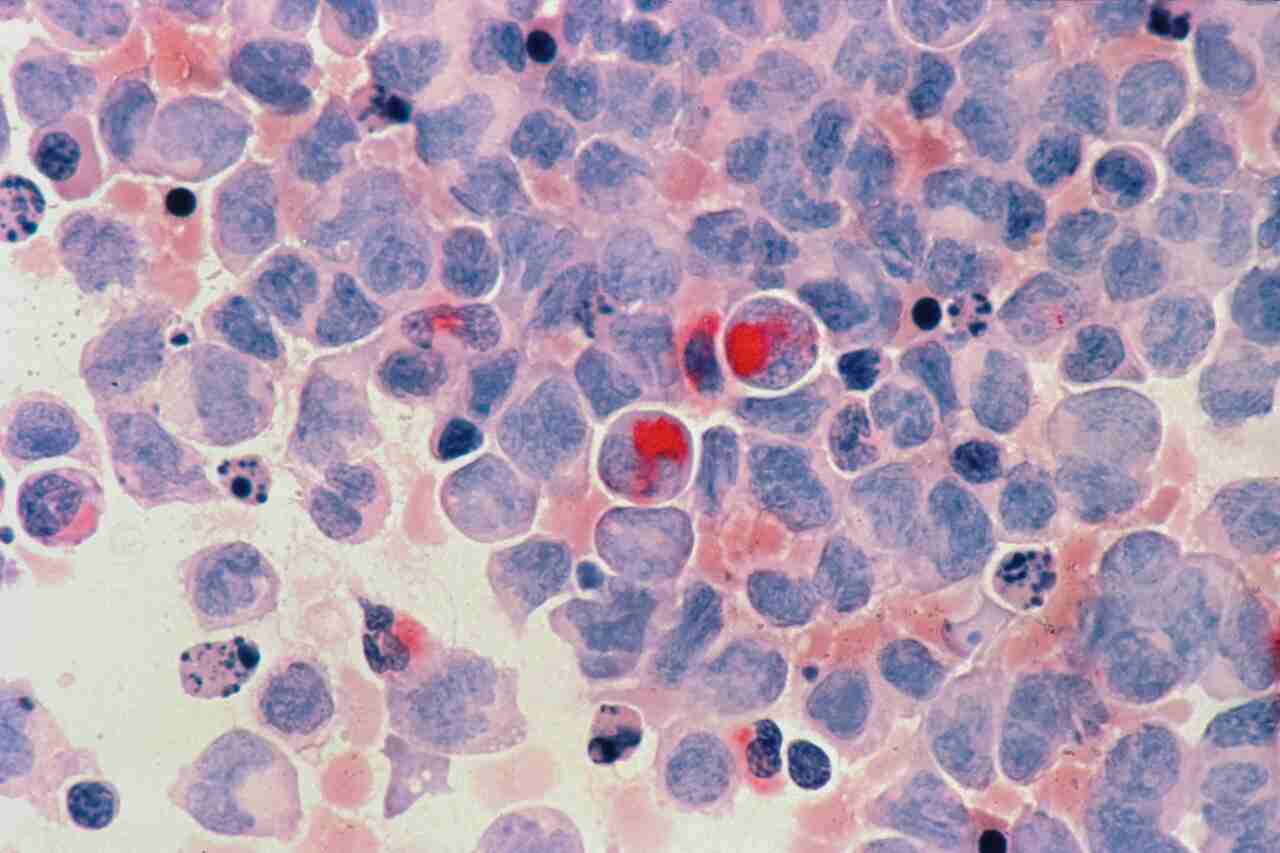
What they found in the blood-forming stem cells blew them away. MLKL wasn’t acting like a straightforward executioner protein. When activated, it slipped right into the mitochondria—those powerhouse organelles that generate the cell’s energy. There, it wreaked havoc: messing up their structure, dropping membrane potential, and crippling energy output. The cell survived, but it started behaving like an aged version of itself, way ahead of schedule.
And that’s the real breakthrough here. We’re not talking mass cell die-off. It’s more like gradual internal breakdown that chips away at performance bit by bit. Picture an engine that’s still firing but sputtering because its core components are wearing out unevenly.
Without MLKL, Blood Stem Cells Held Up Much Better
Knocking out the MLKL protein or blocking its action made a huge difference for the stem cells. They hung onto their self-renewal powers better, churned out healthier immune cells, and shrugged off DNA damage more effectively than cells where MLKL was still calling the shots. Their mitochondria stayed rock-solid, even under stress or in older animals.

The cool part? These improvements didn’t stem from massive gene expression shifts. That points to MLKL directly sabotaging the cell’s hardware—the structures it relies on for peak performance, rather than rewriting its genetic blueprint. In essence, the issue isn’t faulty DNA instructions; it’s the cell’s inner workings grinding to a halt.
Researchers Uncover a Key Driver of Blood System Aging
This finding ties together multiple cellular stresses into one shared culprit: busted mitochondria. It’s not just about the specific stressor hitting the cells. Be it inflammation, DNA glitches, or other age-related hits, they all funnel through MLKL activation, which hammers the mitochondria and drags down blood stem cells.
Why does this hit home? These stem cells are ground zero for blood production. When they degrade, it doesn’t just crimp blood output—it hampers the body’s recovery and infection-fighting prowess too.
“In the long term, this research could lead to therapies that preserve the function of hematopoietic stem cells. That could ultimately improve recovery and long-term health in patients undergoing chemotherapy, radiation, or transplantation,” Dr. Yamashita adds.
That said, heads up: this is mouse-based work unpacking a biological pathway, not a human cure-all yet. Still, it’s a solid leap. If we understand what’s eroding these cells, we might one day hit pause on the process before blood and immunity tank too hard.
MLKL Isn’t Killing Blood Stem Cells—It’s Speeding Up Their Decline
The study, out in Nature Communications, reveals MLKL doesn’t work in blood-forming stem cells quite like we thought. Rather than sparking death, it trashes their mitochondria and slowly saps their strength. The cells stick around, but they can’t operate at full throttle.
That could finally explain why our blood and immune systems fade with age.