Your immune cells do not always lose the fight against cancer because they are too weak. Sometimes, the problem starts much earlier and much deeper, inside the cell itself. T cells, the immune cells that are supposed to find and destroy tumors, face enormous pressure during a long battle with cancer. When that pressure lasts too long, the cell does not simply fail on the outside. First, its internal order begins to fall apart.
Researchers at the University of California San Diego have now shown that T cell exhaustion may be linked to a damaged system that normally sorts and removes faulty proteins inside the cell. In other words, an immune cell does not become passive simply because it “runs out of strength.” Biological waste begins to pile up inside it, waste that should have been broken down and reused. That internal chaos may be one reason the cell eventually stops attacking a tumor effectively.
When the Immune Cell’s Internal Cleanup System Starts to Break Down
A T cell fighting a tumor is not only carrying out an attack. Inside, it is constantly making, folding, repairing, and breaking down proteins that it needs to function. You can think of proteins as tiny working tools inside the cell. When they are damaged or folded the wrong way, they need to be removed. Otherwise, they start creating problems.
This is where the trouble appears to begin. During a long fight against cancer, this internal quality-control system can start to collapse. The T cell may still exist on the outside, but inside it, damaged and misfolded proteins begin to accumulate. The cell can no longer deal with them properly.
“We found that exhausted T cells’ recycling programs are falling apart, leading to damaged and misfolded proteins that pile up with nowhere to go.” Nicole Scharping, University of California San Diego
The result is not the immediate death of the cell, but a gradual loss of function. The T cell enters a state scientists call exhaustion. It no longer attacks as effectively as it once did, loses part of its original power, and gives the tumor room it should not have.
Scientists Identified Three Enzymes That May Help Exhausted T Cells Restore Their Internal Recycling
The researchers therefore looked deeper inside the T cells themselves. Using mass spectrometry, they were able to track thousands of proteins at once and see what was actually happening inside the cell. Not just which genes were active, but what was physically building up or missing. That is where they found a problem that resembles a broken sorting line.
A key role in this system is played by enzymes known as E3 ligases. Their job is to tag damaged or unnecessary proteins so the cell knows they should be sent for breakdown. It is a bit like a label on a package that tells the delivery system where it should go. If that label is missing, the damaged protein stays inside the cell and adds to the internal stress.
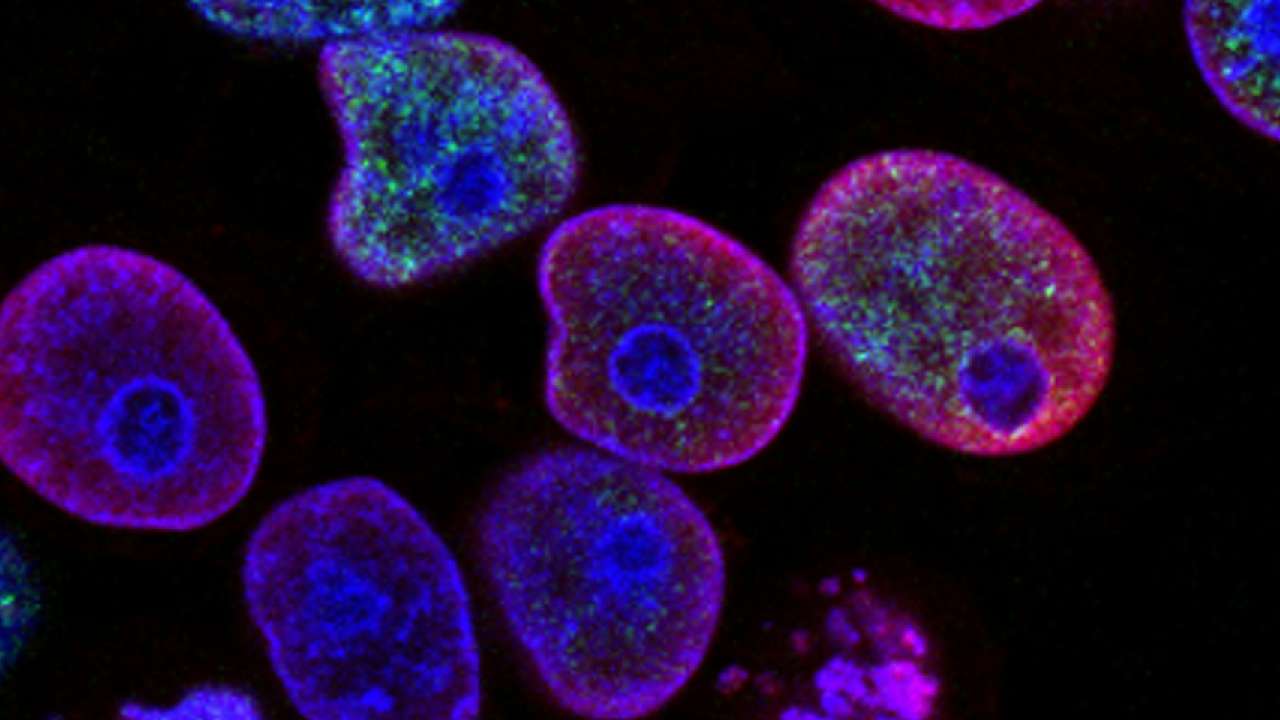
In exhausted T cells, the researchers found that some of these protein “taggers” were no longer working properly. Their attention eventually focused on three specific E3 ligases called NEURL3, RNF149, and WSB1. When the researchers restored them in the cells, the buildup of damaged proteins decreased and the T cells began to perform their job better again.
“When we restored specific E3 ligases, the buildup cleared, and the T cells regained their function and worked better at clearing tumors.” Nicole Scharping, University of California San Diego
Put simply, a T cell does not fail only at the moment when it stops attacking a tumor. The problem may begin earlier, at the level of its own maintenance system. If damaged proteins do not get the correct signal for disposal, they remain inside the cell and increase pressure on the entire system. The cell may still look like a fighter of the immune system, but its internal machinery has already started working against it.
Exhausted T Cells Resemble Neurons in Alzheimer’s Disease. The Problem Is Damaged Proteins Building Up Inside the Cell
This finding is especially interesting because this type of problem is not limited to the immune system. When a cell loses control over its own proteins, it is not a minor defect. Proteins can begin to clump together, misfold, and behave like broken parts that block the entire mechanism instead of being repaired or removed.
Professor Ananda Goldrath points out that exhausted T cells resemble neurons in this respect in diseases such as Parkinson’s and Alzheimer’s. That does not mean these are the same diseases. The similarity lies in the underlying principle. A cell comes under stress, fails to clear damaged proteins, and its function begins to break down.
“We think this loss of proteostasis resembles what occurs in neurons in other protein aggregate diseases such as Parkinson’s and Alzheimer’s. Rescuing these cells from exhaustion could improve the ability of T cells to respond to both chronic infection as well as tumors.” Ananda Goldrath, University of California San Diego
This parallel makes the research more interesting than the discovery of one isolated immune mechanism. It suggests that very different diseases may share a deeper cellular problem. Cells can switch on defensive responses, but still fail to return to balance. In T cells, that means that instead of fighting a tumor for the long haul, they may become trapped in a state they cannot escape without help.
Future Immunotherapies May Need to Help T Cells Last Longer Inside Tumors
The biggest significance of this discovery is not that scientists already have a finished treatment in hand. Rather, they have found a weak point that may help determine whether a T cell survives inside a tumor or gradually breaks down. If its internal protein-control system can be kept running, the cell may be less likely to slide quickly into exhaustion.
This could matter most for therapies that use a patient’s own immune cells. A typical example is CAR T cell therapy. In these treatments, doctors modify immune cells outside the body so they can recognize cancer more effectively, then return them to the patient to attack the tumor in a more targeted way. The problem is that the tumor environment is extremely demanding. Even a cell that has been “armed” in a lab can eventually hit its limits if damaged protein waste starts accumulating inside it.
For now, however, this is not a procedure doctors can simply move into hospitals. The results come mainly from experiments in mice, and there is still a major gap between such a finding and a treatment for people. Still, the researchers have shown that T cell exhaustion may not be only about the tumor itself or a weak immune response. Part of the problem may lie in the cell’s internal maintenance. Future immunotherapies may therefore need to focus not only on directing T cells more strongly against cancer, but also on keeping them stable enough to survive that fight.
Goldrath describes the finding as a target that could be used to design future interventions:
“We uncovered one of the mechanisms by which T cells lose their ability to fight cancer and now we have a target that we can use to design interventions to improve responses to immunotherapy as well as prevent chronic infection.” Ananda Goldrath, University of California San Diego